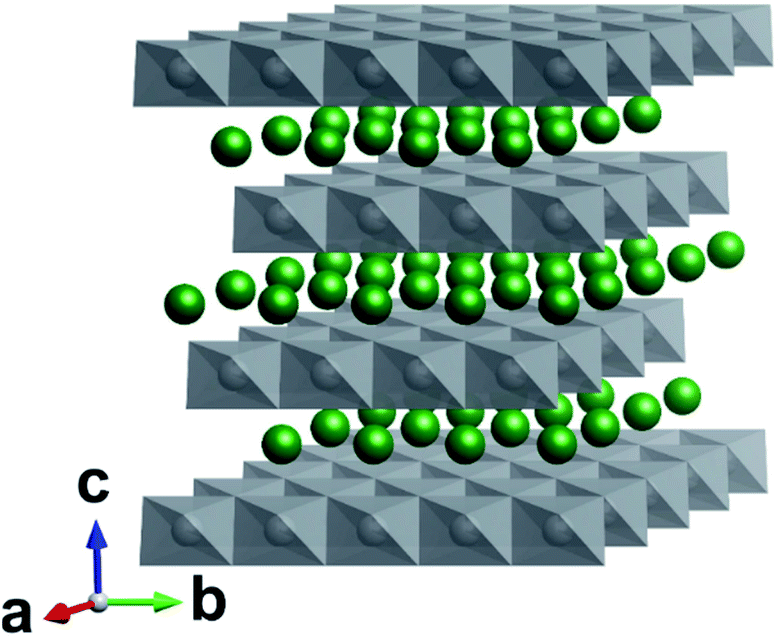 
The cathodic and anodic reactions are presented below. On the anode side, ions begin to intercalate into the anode material, which typically is graphite. As a potential is applied to charge the battery, these ions move out of the structure into the electrolyte. In the discharged state, the cathode material has all of the lithium stored within its layered structure. The principle chemical reaction in a LIB is comparatively simple. The future development of the material is intriguing since it could offer a similar practical capacity to NMC, while other Co-free cathode materials on the market promise capacities similar to LCO. A considerable number of papers have been published on lithium nickel manganese iron oxide ( LNMF or NMF), which have demonstrated that the material could be a viable option for a Co-free cathode material for high energy density LIBs. Additionally, it has a similar size to cobalt, which allows it to replace cobalt in the crystalline lattice with relative ease. Among many elements, iron is an attractive replacement due to its abundance, low cost and relative safety. Thus, the replacement of cobalt in NMC with another transition metal could decrease cost, supply dependance, and toxicity. Furthermore, cobalt is an expensive mineral, which drives up the cost of LIBs. Additionally, the concentration of 70 % of cobalt production in such a small area poses a sever supply risk in case of a crisis. There mining is done in poor conditions, child labor is widely used, the local population is paid poorly for their work and the environment is polluted by the toxic cobalt. Cobalt mining is concentrated in the Democratic republic of Congo, with about 70% of the annual cobalt extraction. The trend in NMC research has been to decrease the quantity of cobalt due to several factors. It possesses an impressive capacity ranging from around 160 mAh/g to 200 mAh/g depending on its composition. Most notably, lithium nickel manganese cobalt oxide (LNMC or NMC) in varying compositions is one of the most widely used battery materials on today’s market. Several competing battery chemistries have since emerged to address performance and stability issues. This was mainly due to irreversible structural changes at higher charging potentials. Though LCO was at its time a revolutionary material, it still suffered from a relatively low specific capacity of only 140 mAh/g (theoretical 274 mAh/g). The development of battery materials has ever since been in rapid increase and several superior battery chemistries have replaced LCO nearly completely. In 2019 Whittingham, Mizushima and Goodenough were awarded with a Nobel prize for their efforts in battery research. The work lead eventually to Sony’s commercialization of the first lithium-ion battery (LIB) in the early 1990s. The next generation cathode material, lithium cobalt oxide (LiCoO 2, LCO), was produced by John Goodenough and Koichi Mizushima in the beginning of the 1980s. This diffusion mechanism was later deemed the intercalation process. The material was based on a process in which lithium ions diffused in between TiS 2 layers. Stanley Whittingham demonstrated the use of TiS 2 as a cathode material. The revolution of electrochemical energy storage took place in the 1970s, when M.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
